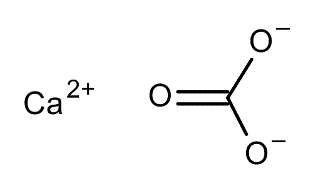
CAL-AID® – Calcium Carbonate BP
COMPOSITION
Calcium Carbonate 500 mg
INDICATIONS
CAL-AID® is used for the treatment or prevention of calcium depletion in patients in whom dietary measures are inadequate. Conditions that may be associated with calcium deficiency include hypoparathyroidism, achlorhydria, chronic diarrhea, vitamin D deficiency, steatorrhea, sprue, pregnancy and lactation, menopause, pancreatitis, renal failure, alkalosis, and hyperphosphataemia. CAL-AID® is being used increasingly often to treat hyperphosphataemia in chronic renal failure as well as those on continuous ambulatory peritoneal dialysis (CAPD) and haemodialysis. Many patients are unable to tolerate sufficient doses for complete phosphate control and require additional measures such as stringent dietary phosphate restriction or relatively small doses of aluminium hydroxide. CAL-AID® containing preparations can provide short-term relief of dyspeptic systems but are no longer recommended for long-term treatment of peptic ulceration.
PHARMACOLOGY
Calcium carbonate reacts with gastric acid to produce a salt and water. For calcium carbonate the postulated chemical reaction is: CaCO3+2HCl = CaCl2+H2O+CO2. Two grams of calcium carbonate will readily bring 100 ml of hydrochloric acid to a pH above 6. The increase in gastric pH diminishes the activity of pepsin in the gastric secretion. Up to 30% of the oral calcium load may be absorbed.
DOSAGE & ADMINISTRATION
CAL-AID® 500 mg tablet: Calcium Carbonate is always used orally and when used as an antacid the recommended doses for adults are equivalent to 540-2000 mg Calcium Carbonate per day, doses for children being half of those for adults. As a dietary supplement, such as for the prevention of osteoporosis, 1250-3750 mg Calcium Carbonate (500-1500 mg calcium) daily is recommended in general, but again this will need to be tailored to the individual patient depending on any specific disease such as Calcium deficiency, malabsorption or parathyroid function. In pregnancy and lactation, the recommended daily dose of calcium is 1200-1500 mg. In chronic renal failure the doses used vary from 2.5-9.0 gm Calcium Carbonate per day and need to be adjusted according to the individual patient. To maximize effective phosphate binding in this context the Calcium Carbonate should be given with meals.
CONTRAINDICATIONS
Hypercalcaemia and hyperparathyroidism
Hypercalciuria and nephrolithiasis
Zollinger-Ellison syndrome
Concomitant digoxin therapy (requires careful monitoring of serum calcium level)
When hypercalcaemia occurs, discontinuation of the drug is usually sufficient to return serum calcium concentrations to normal. Calcium salts should be used cautiously in patients with sarcoidosis, renal or cardiac disease, and in patients receiving cardiac glycosides.
SIDE EFFECTS
Orally administered Cal-aid may be irritating to the GI tract. It may also cause constipation. Hypercalcaemia is rarely produced by administration of calcium alone, but may occur when large doses are given to patients with chronic renal failure.
PREGNANCY & LACTATION
Calcium containing drugs have been widely used in pregnancy by way of oral calcium supplementation or antacid therapy. Calcium Carbonate can be used in lactating women too.
OVERDOSAGE
Symptoms of overdose may include severe nausea, vomiting, dizziness, convulsions and coma after apparent recovery. In case of overdose, one should seek emergency medical attention.
DRUG INTERACTION
CAL-AID® may enhance the cardiac effects of digoxin and other cardiac glycosides, if systemic hypercalcaemia occurs. Cal-aid may interfere with the absorption of concomitantly administered tetracycline preparations and in chronic renal failure modification of vitamin D therapy may be required to avoid hypercalcaemia when CAL-AID® is used as the primary phosphate binder.
STORAGE
Store in a cool, dry place in controlled room temperature.
PACKING
CAL-AID®: Each box contains 10×10 tablets in pack.