L-SOL®–Levosalbutamol INN
COMPOSITION
Available in 2 dosage forms
L-Sol Tablet: Each tablet contains Levosalbutamol Sulphate INN equivalent to Levosalbutamol 2 mg.
L-Sol Syrup: Each 5 ml syrup contains Levosalbutamol Sulphate INN equivalent to Levosalbutamol 1 mg.
PHARMACOLOGY
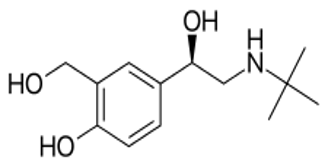
Levosalbutamol is a single isomer beta-2 agonist that differs from racemic salbutamol by elimination of S-salbutamol. L-Salbutamol is an effective bronchodilator whose primary mechanism of action is unimpeded by S-Salbutamol. Thus, when compared with racemic salbutamol, clinically comparable bronchodilation can be achieved with doses which causes substantially lessen beta-mediated side effects.
INDICATIONS
Levosalbutamol is indicated for the treatment or prevention of bronchospasm in adults, adolescents, and children 6 years of age and older with reversible obstructive airway disease.
DOSAGE AND ADMINISTRATION
Tablet-
Adults and children above 12 years: 1-2 mg three times daily. Children (6 -11 years): 1 mg three times daily.
Syrup-
Adults and children above 12 years: 5-10 ml three times daily. Children (6 -11 years): 5 ml three times daily.
SIDE EFFECTS
Potentially serious side effects like hypokalamia may result from beta-2 agonist therapy. This effect may be potentiated by hypoxia. The other likely side effects are gastrointestinal disturbances such as nausea, vomiting, burning substernal or epigastric pain and diarrhoea. In some cases nervousness, headache, dizziness, fatigue and sleeplessness may occur.
PRECAUTIONS
Particular caution is advised in acute severe asthma as this effect may be potentiated by hypoxia and by concomitant treatment with xanthine derivatives, steroids and diuretics. Serum potassium levels should be monitored in such situations. Oral Levosalbutamol should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias or hypertension. Levosalbutamol can be used with caution after myocardial infarction.
CONTRAINDICATIONS
Hypersensitivity to any of the components of the formulation.
DRUG INTERACTIONS
Other short-acting sympathomimetic bronchodilators or epinephrine should be used with caution with Levosalbutamol. If additional adrenergic drugs are to be administered by any route, they should be used with caution to avoid deleterious cardiovascular effects.
USE IN PREGNANCY AND LACTATION
Pregnancy category “C”.
Use in pregnancy: Use of oral Levosalbutamol in pregnant women should be considered only if the expected benefit to the mother is greater than any possible risk to the foetus or the infant.
Use in lactation: It is not known whether Levosalbutamol is excreted in human milk. Caution should be exercised when oral Levosalbutamol is administered to a nursing woman.
OVER DOSAGE
The expected symptoms with overdosage are those of excessive beta-adrenergic stimulation and/or occurrence or exaggeration of any of the symptoms listed under side effects e.g., tachycardia, nervousness, headache, tremor, nausea, dizziness, fatigue and sleeplessness. Hypokalaemia also may occur. Treatment consists of discontinuation of oral Levosalbutamol together with appropriate symptomatic therapy. In the event of serious poisoning, the stomach should be emptied and, if necessary, a beta-blocker administered with caution in patients with a history of bronchospasm.
STORAGE
Store tablet and syrup at or below 30°C. Protect from light & moisture. Keep out of the reach of children.
PACKING
L-SOL®: Tablet 2 mg: Each box contains 10 x10’s tablets in blister pack.
L-SOL®: Syrup 1mg/ 5ml: Each bottle contains 60 ml syrup.