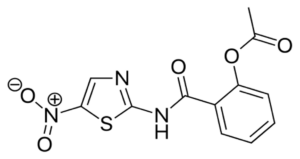
NIZ® – Nitazoxanide INN
COMPOSITION
Available in 2 dosage forms
Niz Tablet: Each film coated tablet contains Nitazoxanide INN 500 mg.
Niz 30 ml suspension: Each 5 ml suspension contains Nitazoxanide INN 100 mg. Niz 60 ml suspension: Each 5 ml suspension contains Nitazoxanide INN 100 mg.
PHARMACOLOGY
Nitazoxanide, a synthetic antiprotozoal agent. Nitazoxanide is well absorbed from GIT. It interferes with the Pyruvate Ferredoxin Oxidoreductase (PFOR) enzyme dependent electron transfer reaction which is essential to anaerobic glucose energy metabolism. This results in cell swelling, membrane damage resulting in dysfunction of the parasite.
INDICATION
Diarrhoea caused by Cryptosporidium parvum and Giardia lambila. Amebiasis and helminth infections
DOSAGE & ADMINISTRATION
Age 1-3 years: 5 ml (100 mg) twice daily for 3 days. Age 4-11 years: 10 ml (200 mg) twice daily for 3 days.
Age > 12 Years 25 ml or 1 tablet (500 mg) twice daily for 3 days. The suspension or tablet should be taken with food.
SIDE EFFECT
Nitazoxanide is generally well tolerated. However abdominal pain. Diarrhoea, vomiting and headache have been reported rarely.
PRECAUTION
Nitazoxanide should be administered with caution to patients with hepatic, renal and biliary disease.
CONTRAINDICATION
Known hypersensitivity to Nitazoxanide of any other ingredient in the formulations.
DRUG INTERACTION
Nitazoxanide is highly bound to plasma protein (>99.9%). Therefore, caution should be used when administering Nitazoxanide concurrently with other highly plasma-protein bound drugs.
USE IN PREGNANCY & LACTATION
Pregnancy category B: This drug should be used during pregnancy only if clearly needed. Nursing mother: It is not known whether Nitazoxanide is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Nitazoxanide is administrated to a nursing mother.
OVER DOSAGE
Information on Nitazoxanide over dosage is not available. Single oral doses up to 4000 mg Nitazoxanide have been administered to healthy adults without significant adverse effects.
RECONSTITUTION FOR SUSPENSION
Niz 30 ml suspension: Shake the bottle well before adding water. Then add 20 ml of boiled and cooled water (with the help of provided cup) into the bottle and shake well to make 60 ml suspension.
STORAGE
Store in a cool and dry place, away from light and moisture. Once reconstituted suspen¬sion should be used within 7 days.
PACKAGING
Niz tablet: Box contains 3 x6 tablets in Alu-Alu blister.
Niz 30 ml suspension: Bottle containing dry powder to reconstitute 30 ml suspension.
Niz 60 ml suspension: Bottle containing dry powder to reconstitute 60 ml suspension.